Translate this page into:
Endoxifen for the management of bipolar disorder with comorbid substance use disorder: A report of two cases

*Corresponding author: Natarajan Shanmugasundaram, Department of Psychiatry, Sri Ramachandra Medical College and Research Institute, Chennai, Tamil Nadu, India. natshanmugasundaram4@gmail.com
-
Received: ,
Accepted: ,
How to cite this article: Dave M, Shanmugasundaram N. Endoxifen for the management of bipolar disorder with comorbid substance use disorder: A report of two cases. Arch Biol Psychiatry 2023;1:25-8.
Abstract
Bipolar disorder (BI) is a common occurrence and increases the risk of substance use disorder. This comorbidity has potentially serious consequences including suicide attempts, hospitalization, and rapid cycling. Endoxifen could play a role in the management of BI based on the inhibition of protein kinase C, which also has a role in substance abuse. We present two cases of patients with bipolar mania and comorbid substance use disorder, who were effectively and safely treated with adjunct Endoxifen. The addition of Endoxifen addition reduced manic symptoms as well as substance use. Furthermore, the inclusion of Endoxifen permitted the use of lower doses of other drugs in one patient and reduced the pill burden in the other patient. Endoxifen has the potential for use in the management of BI in patients with comorbid substance abuse.
Keywords
Bipolar disorder
Endoxifen
Protein kinase
Alcohol use disorder
Substance use disorder
INTRODUCTION
Bipolar disorder (BI) is a challenging presentation in psychiatry and the incidence of comorbid addictions complicates its management. Studies have reported lifetime prevalence rates of comorbidities of BI of up to 39.1% for alcohol abuse, 23.2% for alcohol dependence, 46% for cannabis use, 24% for cocaine use, and 8.5% for opioid use. Comorbid alcohol consumption in patients with bipolar I disorder is associated with suicide attempts and rapid cycling.[1] Tobacco use in BI is associated with rapid cycling, more effective episodes, and a longer duration of hospitalization.[2] We describe the role of Endoxifen in the management of two cases of BI in individuals with comorbid substance use disorder.
CASE SERIES
Case 1
A 35-year-old male with no history of depressive or obsessive-compulsive symptoms or any other psychiatric illness reported experiencing a first manic episode 2–3 weeks prior. The patient was irritable with occasional outbursts of violent anger, excessive grandiose talks, unnecessary spending, poor sleep, increased alcohol consumption and smoking throughout the day.
The patient had a history of alcohol consumption for over 15 years and consumed eight to ten pegs of whiskey daily for 6–7 months. He smoked 20 cigarettes daily and made no attempts to abstain from smoking or alcohol. His father and brother also consumed alcohol, while his mother was being treated for depression. The patient was married and belonged to the middle socioeconomic status.
The patient was diagnosed with bipolar I disorder, alcohol use disorder, and nicotine use disorder. Investigations revealed normal hematology, elevated alanine aminotransferase, elevated aspartate aminotransferase, elevated gamma-glutamyl transferase, normal findings on ultrasonography of the abdomen, normal echocardiography, and magnetic resonance imaging of the brain. The CGI score has been added using the Insert tool 6.[3]
The patient was hospitalized and was prescribed thiamine and multivitamins for alcohol withdrawal. Lorazepam 12–14 mg daily was initiated. The patient was prescribed Tab. Endoxifen 8 mg once daily and Tab. Olanzapine 15 mg daily in divided doses. Intramuscular haloperidol and promethazine were reserved for emergencies, as needed.
After 10 days, there was complete remission of manic symptoms and alcohol withdrawal. The CGI score has been added using the Insert tool 1. The patient was prescribed Endoxifen 8 mg once daily, Olanzapine 10 mg/day in two divided doses, Lorazepam 2 mg once daily at bedtime, and multivitamins once daily. Remission was maintained at 2 weeks post-discharge and the patient remained sober. CGI Score at 1 and 2 weeks was 1. CGI global improvement at 1 and 2 weeks was 1 while CGI efficacy index at 1 and 2 weeks was 1.
At 33 days after presentation, the patient was stable on Endoxifen 8 mg once daily, Olanzapine 5 mg twice daily, Lorazepam 2 mg at bedtime, and multivitamins once daily. CGI severity score was 1, CGI global improvement was 1 [Figure 1], and CGI efficacy index was 1. No side effects were reported. The patient was adherent to therapy. Vegetative functions had normalized and the patient reported minimal craving for alcohol and nicotine during 42 days of Endoxifen use. The treatment details for Case 1 are described in Table 1.
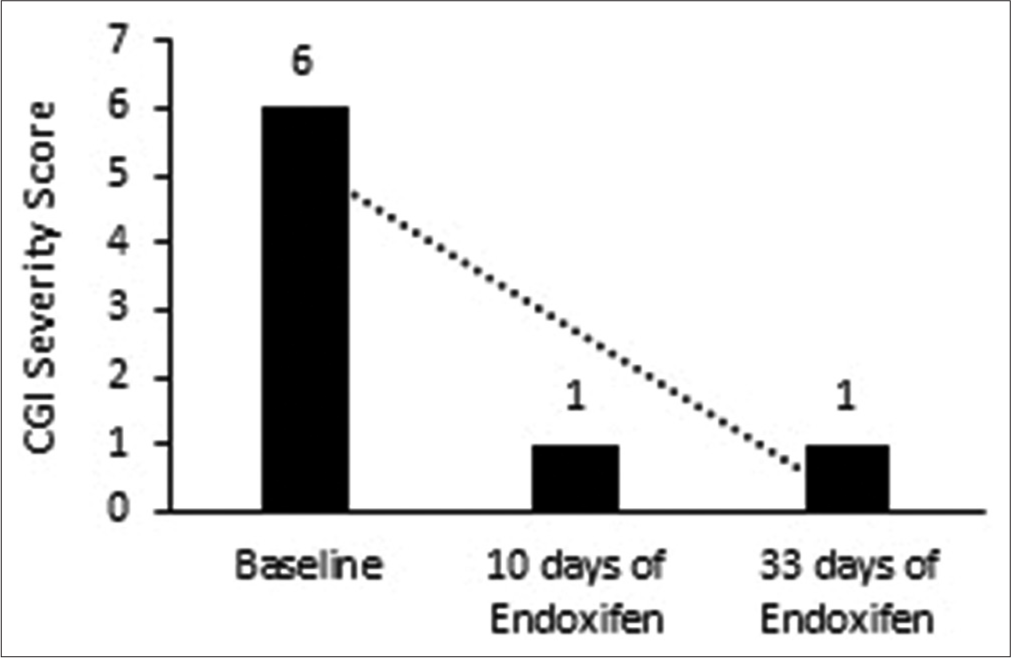
- Change in clinical global impression severity score for case 1.
| Parameters | Case 1 | Case 2 |
|---|---|---|
| Age Gender Substance use Setting Drugs given along with Endoxifen at beginning Endoxifen therapy planned as Response with Endoxifen Drugs given along with Endoxifen after seeing response Duration of Endoxifen treatment Substance usage during Endoxifen treatment |
35 years Male Alcohol, nicotine Inpatient first, followed by outpatient Thiamine and multivitamins, Lorazepam 12–14 mg daily, Endoxifen 8 mg OD and Olanzapine 15 mg daily in divided doses; intramuscular haloperidol and promethazine reserved for emergency as needed Primary therapy At 10 days Olanzapine 5 mg BID, Lorazepam 2 mg at bedtime and multivitamins OD 42 days Patient reported minimal craving for alcohol and nicotine during Endoxifen treatment |
30 years Male Cannabis, nicotine, alcohol Outpatient Sodium valproate 750 mg BID (cross-tapered with Endoxifen), Olanzapine 20 mg at bedtime, clonazepam 1 mg at night for 3 months, lithium 300 mg TDS Sequential therapy At 3 days Olanzapine 20 mg at bedtime, clonazepam 1 mg, lithium 300 mg TDS 3 months Patient did not resume substance usage with Endoxifen treatment |
BID: Twice daily, HS: At night, OD: Once-daily, TDS: Three times a day
Case 2
A 30-year-old male taxi driver presented with complaints of lack of sleep for 1 week, wandering during the night, abusing his family members, and making tall claims. The patient had a history of use of cannabis, nicotine, and alcohol. The patient was married. The CGI score has been added using the Insert tool 6.
The patient was diagnosed with the bipolar affective disorder with comorbid substance use disorder. The patient was prescribed sodium valproate 750 mg twice daily (cross-tapered with Endoxifen later), Olanzapine 20 mg nightly, Clonazepam 1 mg nightly for 3 months, and Lithium 300 mg thrice daily.
There was no clinical improvement after 4 weeks, with minimal change in YMRS scores. The patient denied undergoing electroconvulsive therapy. Therefore, Endoxifen 8 mg once daily in the morning was initiated. After 1 week, the patient showed clinical improvement and change in YMRS ratings. Sodium valproate was tapered and stopped.
Remission began 1 week after initiating Endoxifen treatment. Symptom severity was reduced, as indicated by a CGI score of 2. CGI score was one at the end of 3 months [Figure 2]. No side effects attributed to Endoxifen were observed. Mild rigidity due to concomitant antipsychotic medication was reported, which resolved with 2 weeks of Trihexyphenidyl treatment. The patient resumed his driving job and did not resume substance usage with Endoxifen treatment at 3 months of follow-up. The treatment details for Case 2 are described in Table 1.
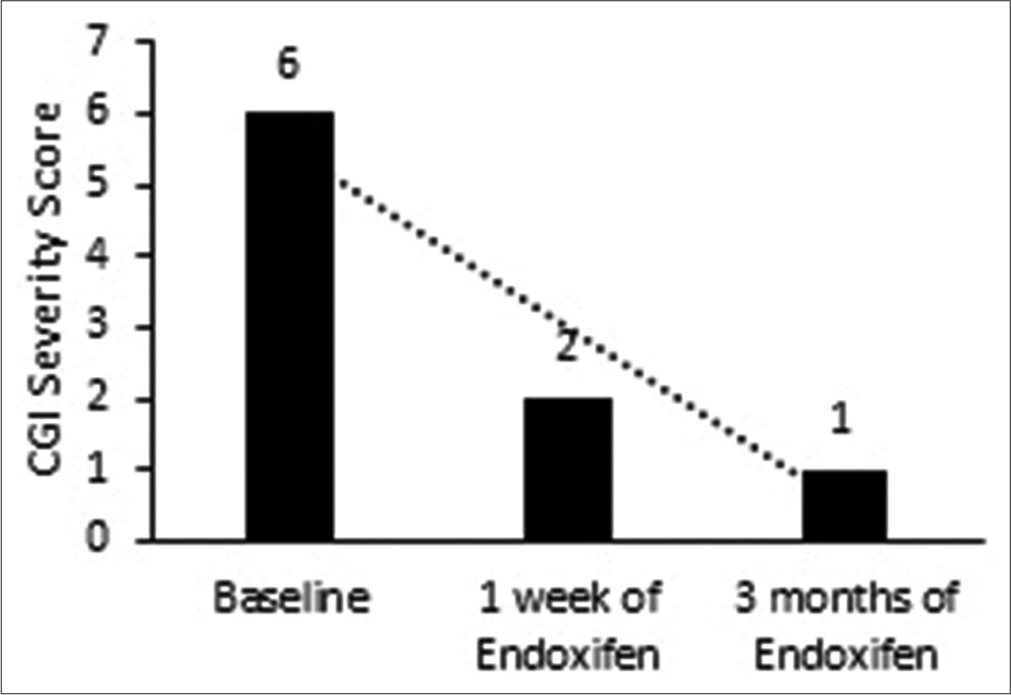
- Change in clinical global impression severity score for case 2.
DISCUSSION
The cases described above highlight the efficacy of Endoxifen in combination with other medications for managing bipolar mania with comorbid substance abuse. In the first case, Endoxifen as part of combination therapy allowed for the reduction of the dosage of other drugs. Olanzapine was initiated as it is a first-line atypical antipsychotic agent, while Endoxifen was initiated based on its utility for managing mania. In the second case, Endoxifen was added to mood stabilizers, Olanzapine, and clonazepam due to a lack of improvement with the latter, leading to rapid improvement of symptoms. It reduced the pill burden and the need for other mood stabilizers. In both cases, Endoxifen effectively and safely reduced manic symptoms and substance usage. Endoxifen was a useful part of pharmacotherapy, leading to mood stabilization, improved symptoms, and reduced substance use or prevented resuming use of a substance(s). Endoxifen may help in the long-term management of alcohol and other substance use disorders.
Since comorbid alcohol use disorder and BI have a poor prognosis,[1] the management strategy requires interventions (pharmacological and psychosocial) that depend on the severity of the addiction.[4] Guidelines published by the National Institute for Health and Care Excellence in 2020 describe the use of several drugs for BI. Referral to secondary care is recommended for patients with comorbid substance use disorder, and such comorbidities require appropriate therapy.[5]
Protein kinase C (PKC) as a common thread
There may be a shared mechanism between BI and addictions,[4] possibly regulated by PKC. Overactive PKC intracellular signalling is associated with mania. Endoxifen inhibits PKC up to 80% and could, thus, play a key role in managing BI.[6] Furthermore, chronic exposure to ethanol increases the activity of PKC and the expression levels of PKC isozymes. A PKC isotype is involved in nicotine addiction.[3,7,8] Therefore, Endoxifen, a PKC inhibitor, could play an important therapeutic role.[6]
In vitro studies have demonstrated that chronic exposure to ethanol increases the activity of PKC[7] and it is suggested that selective inhibition of PKC epsilon could reduce excessive consumption of alcohol.[9] Concerning nicotine addiction, PKC epsilon regulates reward signalling via nicotinic receptors, thus indicating the potential for PKC epsilon as a target for treating comorbid nicotine addiction.[8]
Role of Endoxifen
Clinical studies have demonstrated the efficacy of Endoxifen. Treatment with Endoxifen 8 mg/day led to a nearly 60% reduction in YMRS score, similar to that achieved with Tab. Divalproex 1000 mg/day.[3] In patients with bipolar I disorder, Endoxifen, and Divalproex co-treatment for 21 days led to comparable changes in YMRS score, Montgomery-Åsberg Depression Rating Scale score, and CGI CGI-BP ratings. Endoxifen led to earlier remission (by day 4) compared with divalproex; unlike divalproex, Endoxifen did not increase levels of thyroid-stimulating hormone or decrease levels of platelets.[10]
The current case series highlights the benefits of Endoxifen with substances such as cannabis and nicotine. PKC inhibitors such as Endoxifen can be considered a new comprehensive treatment option in mania with comorbid substance abuse, where it can bring dual benefits by controlling both conditions.
CONCLUSION
Our findings indicate the potential of Endoxifen in managing bipolar mania in patients with comorbid substance use disorder. These patients require simultaneous management of both conditions and regular follow-up to ensure adherence to therapy and maintenance of remission. PKC is involved in BI and substance use disorders. Endoxifen, being a direct PKC inhibitor, can allow for comprehensive management of mania with comorbid substance abuse. As part of combination therapy, Endoxifen demonstrated good tolerability and efficacy in both cases by reducing manic symptoms as well as craving for substances such as nicotine, alcohol, and cannabis. The inclusion of Endoxifen also permitted the use of lower doses of other drugs in one patient and reduced the pill burden in the other patient. Along with previous reports, our findings build the case for Endoxifen as part of the comprehensive management of BI. Endoxifen can be safely used in combination with standard pharmacological treatments for bipolar and substance use disorders. It can be used in a daily dose of 8–16 mg, preferably for an extended period (at least 6 months).
Declaration of patient consent
The authors certify that they have obtained all appropriate patient consent.
Conflicts of interest
There are no conflicts of interest.
Financial support and sponsorship
Nil.
References
- Comorbid bipolar and alcohol use disorder-a therapeutic challenge. Front Psychiatry. 2021;12:660432.
- [CrossRef] [PubMed] [Google Scholar]
- Tobacco use in bipolar disorder. Clin Psychopharmacol Neurosci. 2015;13:1-11.
- [CrossRef] [PubMed] [Google Scholar]
- Endoxifen, a new treatment option for mania: A double-blind, active-controlled trial demonstrates the antimanic efficacy of Endoxifen. Clin Transl Sci. 2016;9:252-9.
- [CrossRef] [PubMed] [Google Scholar]
- Advisory: An Introduction to Bipolar Disorder and Co-Occurring Substance use Disorders. Vol 15. North Bethesda: Substance Abuse and Mental Health Services Administration; 2016.
- [Google Scholar]
- Bipolar Disorder: Assessment and Management London: National Institute for Health and Care Excellence (NICE); 2014.
- [Google Scholar]
- Endoxifen is a new potent inhibitor of PKC: A potential therapeutic agent for bipolar disorder. Bioorg Med Chem Lett. 2010;20:2665-7.
- [CrossRef] [PubMed] [Google Scholar]
- Chronic ethanol exposure increases levels of protein Kinase C delta and epsilon and protein Kinase C-mediated phosphorylation in cultured neural cells. J Biol Chem. 1991;266:23428-32.
- [CrossRef] [PubMed] [Google Scholar]
- Protein Kinase C epsilon modulates nicotine consumption and dopamine reward signals in the nucleus accumbens. Proc Natl Acad Sci U S A. 2011;108:16080-5.
- [CrossRef] [PubMed] [Google Scholar]
- Selective chemical genetic inhibition of protein Kinase C epsilon reduces ethanol consumption in mice. Neuropharmacolgy. 2016;107:40-8.
- [CrossRef] [PubMed] [Google Scholar]
- Endoxifen: A new, protein Kinase C inhibitor to treat acute and mixed mania associated with bipolar I disorder. Bipolar Disord. 2021;23:595-603.
- [CrossRef] [PubMed] [Google Scholar]








